|
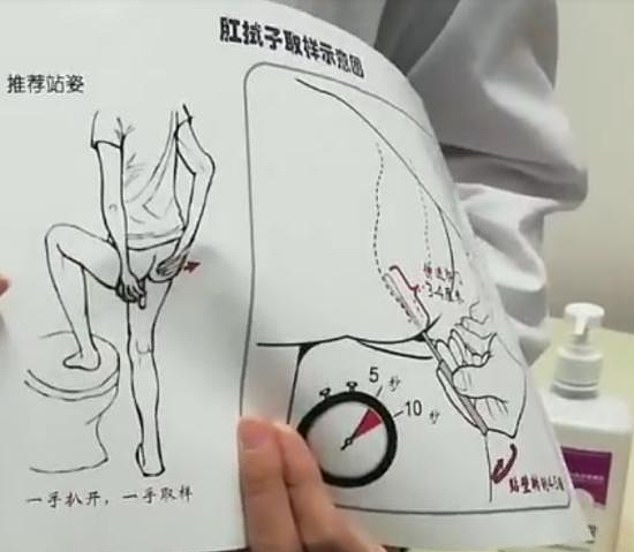
Beijing is using anal swabs to test its residents for coronavirus, a method that experts say is more accurate and raises the chances of detecting the virus.
To collect test samples, the swab needs to be inserted about three to five centimetres (1.2 to 2 inches) into the rectum and rotated several times. After completing the motion twice, the swab is removed before being securely placed inside a sample container. The whole procedure is said to take about 10 seconds. Fifty-five people in the United States have died after receiving a COVID-19 vaccine, according to reports submitted to a federal system. Deaths have occurred among people receiving both the Moderna and the Pfizer-BioNTech vaccines, according to the reports. The reporting system, the Vaccine Adverse Event Reporting System (VAERS), is a federal database. The system is passive, meaning reports aren’t automatically collected and must be filed. VAERS reports can be filed by anyone, including health care providers, patients, or family members. VAERS reports “often lack details and sometimes can have information that contains errors,” according to the reporting system’s website. Still, reports on VAERS represent “only a small fraction of actual adverse events,” the site states, though underreporting is believed to be less common for serious events. In some cases, patients died within days of receiving a COVID-19 vaccine. One man, a 66-year-old senior home resident in Colorado, was sleepy and stayed in bed a day after getting Moderna’s vaccine. Early the next morning, on Christmas Day, the resident “was observed in bed lying still, pale, eyes half open and foam coming from mouth and unresponsive,” the VAERS report states. “He was not breathing and with no pulse.” In another case, a 93-year-old South Dakota man was injected with the Pfizer-BioNTech vaccine on Jan. 4 around 11 a.m. About two hours later, he said he was tired and couldn’t continue with the physical therapy he was doing any longer. He was taken back to his room, where he said his legs felt heavy. Soon after, he stopped breathing. A nurse declared a do-not-resuscitate order. Moderna and Pfizer didn’t respond to requests for comment. Abigail Capobianco, a spokeswoman for the Food and Drug Administration (FDA), told The Epoch Times via email, “Any reports of death following the administration of vaccines are promptly and rigorously investigated jointly by FDA” and the Centers for Disease Control and Prevention (CDC). Officials at the agencies work with health care providers to obtain medical histories and clinical follow-up information and conduct a clinical case review, she said, adding, “It is important to note that it is generally not possible to find out from VAERS data if a vaccine caused an adverse event.” Health officials on the VAERS website caution that a report to the system doesn’t prove a vaccine caused the adverse event, and that no proof that the event was caused by the vaccine is required in order for the system to accept the report. In addition to the deaths, people have reported 96 life-threatening events following COVID-19 vaccinations, as well as 24 permanent disabilities, 225 hospitalizations, and 1,388 emergency room visits.
COVID-19 is the disease caused by the CCP (Chinese Communist Party) virus. Neither the CDC nor the FDA has a central database of reported adverse events. Nancy Messonnier, director of the CDC’s National Center for Immunization and Respiratory Diseases, said on Jan. 6 that severe allergic reactions to COVID-19 vaccines were happening at a rate of 11.1 per million vaccinations, compared to the rate of 1.3 per 1 million flu shots. As of Jan. 15, 10.5 million Americans have received a COVID-19 vaccine. Federal health officials have said that adverse events are being investigated but that the vaccines are still safe to get. “These are safe and effective vaccines. We have good data to show that,” Messonnier said. One death following a vaccination attracted attention earlier this week. Gregory Michael, a 56-year-old medical worker at Mount Sinai Medical Center in Miami, Florida, was injected on Dec. 18, 2020. Sixteen days later, he died. Michael’s wife Heidi Neckelmann said on Facebook that her husband was “very healthy” before being vaccinated. She said he was admitted to an intensive care unit with a diagnosis of acute idiopathic thrombocytopenic purpura, a blood disorder, caused by a reaction to the vaccine. He eventually passed away. “He was a pro-vaccine advocate, that is why he got it himself,” Neckelmann wrote. “I believe that people should be aware that side effects can happen, that it is not good for everyone and in this case destroyed a beautiful life, a perfect family, and has affected so many people in the community. Do not let his death be in vain, please save more lives by making this information news.” Pfizer told news outlets in a statement that no evidence currently shows a link between the death and its vaccine. “Pfizer and BioNTech are aware of the death of a health care professional 16 days after receiving a first dose of BNT162b2,” Pfizer said in a statement. “It is a highly unusual clinical case of severe thrombocytopenia, a condition that decreases the body’s ability to clot blood and stop internal bleeding.” “We are actively investigating this case, but we don’t believe at this time that there is any direct connection to the vaccine,” the company added. “There have been no related safety signals identified in our clinical trials, the post-marketing experience thus far, or with the mRNA vaccine platform. To date, millions of people have been vaccinated and we are closely monitoring all adverse events in individuals receiving our vaccine. It is important to note that serious adverse events, including deaths that are unrelated to the vaccine, are unfortunately likely to occur at a similar rate as they would in the general population.” A Pfizer spokesperson told The Epoch Times via email earlier this month that the company “closely monitor[s] all reports following vaccination.” Moderna hasn’t responded to requests for comment on its involvement in monitoring serious adverse events in people who get its vaccine. The updated VAERS data came after Norway changed its COVID-19 vaccination guide to direct officials not to give “very frail” people one of the vaccines, citing 13 deaths among people who were vaccinated. (THE DAILY MAIL) — Scientists have discovered Australia is in the grips of two different strains of coronavirus.
Cambridge University researchers mapped the genetic history of the infection from December to March and found three distinct, but closely related, variants. While Australian authorities are struggling to find a cure for the original virus which developed from animals, they’re also battling a mutation known as strain C. Type A is the most prevalent in Australia, however Type C has also been recorded in Sydney, according to the experts. Analysis of the strains showed type A – the original virus that jumped to humans from bats via pangolins – was not China’s most common. Instead, the pandemic’s ground-zero was mainly hit by type B, which was in circulation as far back as Christmas Eve. Type B was also the dominant strain across large parts of the United Kingdom and Europe, but has not made it to Australia.Type C was an offshoot of Type B, mutating from the secondary strain and spreading to Europe and Australia via Singapore. Scientists believe the virus – officially called SARS-CoV-2 – is constantly mutating to overcome immune system resistance in different populations. The academics’ published work – which has been scrutinised by fellow scientists – only traced the samples of 160 patients across the world, including many of the first cases in Europe and the US. Methods used to trace the prehistoric migration of ancient humans were adapted to track the spread of the SARS-CoV-2 virus, which causes COVID-19. The team have now updated their analysis to include more than 1,000 COVID-19 cases up to the end of March to provide a clearer snapshot. It has not yet been peer-reviewed. The smaller snapshot, published in the journal PNAS, initially suggested that type C was the most common in Europe. But the data now shows type B is spreading more rampantly – all but one of 31 SARS-CoV-2 samples taken from patients in Switzerland were of the second cluster. Two similar incidents were recorded in initial vaccine recipients in the United Kingdom last week Suffered Anaphylactic reaction - may cause death
https://justthenews.com/politics-policy/coronavirus/alaskan-health-care-worker-suffers-anaphylactic-reaction-pfizer-vaccine
Your Coronavirus Test Is Positive. Maybe It Shouldn’t Be.The usual diagnostic tests may simply be too sensitive and too slow to contain the spread of the virus.
By Apoorva Mandavilli Published Aug. 29, 2020Updated Sept. 17, 2020 https://www.nytimes.com/2020/08/29/health/coronavirus-testing.html Some of the nation’s leading public health experts are raising a new concern in the endless debate over coronavirus testing in the United States: The standard tests are diagnosing huge numbers of people who may be carrying relatively insignificant amounts of the virus. Most of these people are not likely to be contagious, and identifying them may contribute to bottlenecks that prevent those who are contagious from being found in time. But researchers say the solution is not to test less, or to skip testing people without symptoms, as recently suggested by the Centers for Disease Control and Prevention. Instead, new data underscore the need for more widespread use of rapid tests, even if they are less sensitive. “The decision not to test asymptomatic people is just really backward,” said Dr. Michael Mina, an epidemiologist at the Harvard T.H. Chan School of Public Health, referring to the C.D.C. recommendation. “In fact, we should be ramping up testing of all different people,” he said, “but we have to do it through whole different mechanisms.” In what may be a step in this direction, the Trump administration announced on Thursday that it would purchase 150 million rapid tests.
But similar PCR tests for other viruses do offer some sense of how contagious an infected patient may be: The results may include a rough estimate of the amount of virus in the patient’s body. “We’ve been using one type of data for everything, and that is just plus or minus — that’s all,” Dr. Mina said. “We’re using that for clinical diagnostics, for public health, for policy decision-making.” But yes-no isn’t good enough, he added. It’s the amount of virus that should dictate the infected patient’s next steps. “It’s really irresponsible, I think, to forgo the recognition that this is a quantitative issue,” Dr. Mina said. The PCR test amplifies genetic matter from the virus in cycles; the fewer cycles required, the greater the amount of virus, or viral load, in the sample. The greater the viral load, the more likely the patient is to be contagious. This number of amplification cycles needed to find the virus, called the cycle threshold, is never included in the results sent to doctors and coronavirus patients, although it could tell them how infectious the patients are. In three sets of testing data that include cycle thresholds, compiled by officials in Massachusetts, New York and Nevada, up to 90 percent of people testing positive carried barely any virus, a review by The Times found. On Thursday, the United States recorded 45,604 new coronavirus cases, according to a database maintained by The Times. If the rates of contagiousness in Massachusetts and New York were to apply nationwide, then perhaps only 4,500 of those people may actually need to isolate and submit to contact tracing. One solution would be to adjust the cycle threshold used now to decide that a patient is infected. Most tests set the limit at 40, a few at 37. This means that you are positive for the coronavirus if the test process required up to 40 cycles, or 37, to detect the virus. Tests with thresholds so high may detect not just live virus but also genetic fragments, leftovers from infection that pose no particular risk — akin to finding a hair in a room long after a person has left, Dr. Mina said. Any test with a cycle threshold above 35 is too sensitive, agreed Juliet Morrison, a virologist at the University of California, Riverside. “I’m shocked that people would think that 40 could represent a positive,” she said. A more reasonable cutoff would be 30 to 35, she added. Dr. Mina said he would set the figure at 30, or even less. Those changes would mean the amount of genetic material in a patient’s sample would have to be 100-fold to 1,000-fold that of the current standard for the test to return a positive result — at least, one worth acting on.
The Food and Drug Administration said in an emailed statement that it does not specify the cycle threshold ranges used to determine who is positive, and that “commercial manufacturers and laboratories set their own.”
The Centers for Disease Control and Prevention said it is examining the use of cycle threshold measures “for policy decisions.” The agency said it would need to collaborate with the F.D.A. and with device manufacturers to ensure the measures “can be used properly and with assurance that we know what they mean.” The C.D.C.’s own calculations suggest that it is extremely difficult to detect any live virus in a sample above a threshold of 33 cycles. Officials at some state labs said the C.D.C. had not asked them to note threshold values or to share them with contact-tracing organizations. For example, North Carolina’s state lab uses the Thermo Fisher coronavirus test, which automatically classifies results based on a cutoff of 37 cycles. A spokeswoman for the lab said testers did not have access to the precise numbers. This amounts to an enormous missed opportunity to learn more about the disease, some experts said. “It’s just kind of mind-blowing to me that people are not recording the C.T. values from all these tests — that they’re just returning a positive or a negative,” said Angela Rasmussen, a virologist at Columbia University in New York. “It would be useful information to know if somebody’s positive, whether they have a high viral load or a low viral load,” she added. Officials at the Wadsworth Center, New York’s state lab, have access to C.T. values from tests they have processed, and analyzed their numbers at The Times’s request. In July, the lab identified 872 positive tests, based on a threshold of 40 cycles. With a cutoff of 35, about 43 percent of those tests would no longer qualify as positive. About 63 percent would no longer be judged positive if the cycles were limited to 30. In Massachusetts, from 85 to 90 percent of people who tested positive in July with a cycle threshold of 40 would have been deemed negative if the threshold were 30 cycles, Dr. Mina said. “I would say that none of those people should be contact-traced, not one,” he said. Other experts informed of these numbers were stunned. “I’m really shocked that it could be that high — the proportion of people with high C.T. value results,” said Dr. Ashish Jha, director of the Harvard Global Health Institute. “Boy, does it really change the way we need to be thinking about testing.” Dr. Jha said he had thought of the PCR test as a problem because it cannot scale to the volume, frequency or speed of tests needed. “But what I am realizing is that a really substantial part of the problem is that we’re not even testing the people who we need to be testing,” he said. The number of people with positive results who aren’t infectious is particularly concerning, said Scott Becker, executive director of the Association of Public Health Laboratories. “That worries me a lot, just because it’s so high,” he said, adding that the organization intended to meet with Dr. Mina to discuss the issue. The F.D.A. noted that people may have a low viral load when they are newly infected. A test with less sensitivity would miss these infections. But that problem is easily solved, Dr. Mina said: “Test them again, six hours later or 15 hours later or whatever,” he said. A rapid test would find these patients quickly, even if it were less sensitive, because their viral loads would quickly rise. PCR tests still have a role, he and other experts said. For example, their sensitivity is an asset when identifying newly infected people to enroll in clinical trials of drugs. But with 20 percent or more of people testing positive for the virus in some parts of the country, Dr. Mina and other researchers are questioning the use of PCR tests as a frontline diagnostic tool. People infected with the virus are most infectious from a day or two before symptoms appear till about five days after. But at the current testing rates, “you’re not going to be doing it frequently enough to have any chance of really capturing somebody in that window,” Dr. Mina added. Highly sensitive PCR tests seemed like the best option for tracking the coronavirus at the start of the pandemic. But for the outbreaks raging now, he said, what’s needed are coronavirus tests that are fast, cheap and abundant enough to frequently test everyone who needs it — even if the tests are less sensitive. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |








 RSS Feed
RSS Feed
